
Climate Monitoring Innovation Trends: 5 Signals Emerging from Recent Patent Filings
When you hear the term climate monitoring, the picture that usually comes to mind is a network of satellites and machines deployed globally. You imagine

A bioprinter can now lay down living material one precise layer at a time, turning a droplet of bioink into something biological: a steak, a strip of tissue, or even the early shape of an organ.
What seemed fictional at one point is now moving closer to real-world application. And there’s a lot happening in the domain. Researchers are exploring ways to print cartilage, repair damaged tissue, build vascularized systems, and even recreate miniature versions of organs for drug testing. In parallel, the same technology is being explored for everything from regenerative medicine to cultivated meat.
But beneath those headline moments lies a much larger innovation story. Bioprinting is not advancing through a single breakthrough. It is evolving through a dense ecosystem of materials, methods, and biological systems – from bioinks and hydrogels to scaffold architectures and tissue fabrication strategies.
We were curious to see where inventors are concentrating their efforts, so we explored recent patent filings to understand what is really happening under the hood. Here is what we found.
To understand where bioprinting innovation is moving, we explored recent patent filings using PQAI’s search engine, focusing primarily on filings published from January 2024 onwards.
Source – PQAI
We kept the query broad to capture the wider bioprinting ecosystem. From our search, there emerged a surprisingly diverse landscape.
We saw that some inventions focused on improving the mechanics of printing living cells, while others focused on the biological side of the problem, including new hydrogel formulations and scaffold materials designed to support tissue growth after printing.
There was a lot going on, and it was clear that innovation is happening simultaneously at multiple layers of the bioprinting stack. From our analysis, several recurring innovation signals began to emerge. In the next section, we highlight six key trends that appear to be shaping the future direction of bioprinting technologies.
In the early days of bioprinting, bioinks were mostly treated as carriers. Their job was to hold cells together long enough for a printer to deposit them in the right shape.
But recent patent filings suggest that bioinks are becoming far more active than that. The latest filings suggest that these bioinks can now release compounds, guide tissue development, and influence how cells behave after printing. In other words, the ink itself is starting to shape the biological outcome.
One example is US2025283035A1, which describes morphogenic compound-releasing microspheres embedded within a bioink formulation. In simple terms, the ink contains microscopic biodegradable particles that slowly release biological signals after printing, helping stem cells develop into specific tissue types.
We also saw a notable Chinese patent, CN119367608A, which focuses on a photosensitive collagen-based bioink. The idea here is that the material can harden under light during printing while still supporting living cells. This makes it easier to print precise biological structures that remain stable and allow cells to grow and spread inside the printed scaffold.
Another patent, CN119367605A, filed by the same assignee Collagen Wuhan Biotechnology Co., Ltd., introduces Fe³⁺-coordinated collagen bioinks which are designed to produce stronger and more durable printed scaffolds. These scaffolds also encourage endothelial cells to form vascular networks, which is important for tissue healing and regeneration.
There is a shift in thinking. Bioinks are now evolving from simple printable carriers into active materials that help shape how printed tissues grow and function.
You might think that the biggest breakthroughs in bioprinting are happening in printers. But that’s not the case.
There are other critical components too. One of them being the soft material environments that keep printed cells alive. In bioprinting, those components are hydrogels, which are water-rich, gel-like materials that act as the living environment around cells, helping them survive, attach, and grow after printing.
A lot of innovation is happening in this area, too. One example being US2025223568A1, which describes extracellular matrix hydrogels derived from organoids. These materials attempt to recreate the natural cellular environment found inside human tissue, making it easier for printed cells to survive and organize after printing.
There are other fillings too, like US2025082820A1, which forms a stronger and more stable gel that can support implanted cells and help damaged soft tissue regenerate.
Meanwhile, CN118086170A introduces hydrogel systems designed with 4D extracellular matrix behavior, meaning the printed hydrogel structure can change its shape or mechanical properties over time to better match the needs of different tissues.
There is a lot of work happening around hydrogel engineering projects and if you are working in this domain, there is space for more.
While there is a lot of conversation about self-organizing tissues and scaffold-free biology, the current reality is that scaffolds still remain a major part of the picture. Printed tissues often need architectural support to survive, organize, and maintain their structure after fabrication.
That is why so much innovation is still happening at the scaffold layer. For instance, we saw filings like CN119345463A, which focused on making scaffolds easier to print without losing cell compatibility. Whereas others delved on improving what happens after printing, whether that means better mechanical stability, more controlled pore architecture, or higher biological activity.
US2025120800A1, for example, focuses on maintaining scaffold structural integrity in engineered tissues. The patent application addresses real-world issues such as delamination, weakening, and collapse during or after implantation, pointing to a shift from simply printing scaffolds to making sure they remain stable in use.
There are other filings too that show how this layer is becoming more sophisticated. US2025171736A1, for instance, explores a cell-support matrix with highly controlled, vertically organized porosity to support cartilage formation, while CN119215231A describes a bioactive 3D-printed hydrogel scaffold incorporating stem cell exosomes that can help improve tissue formation.
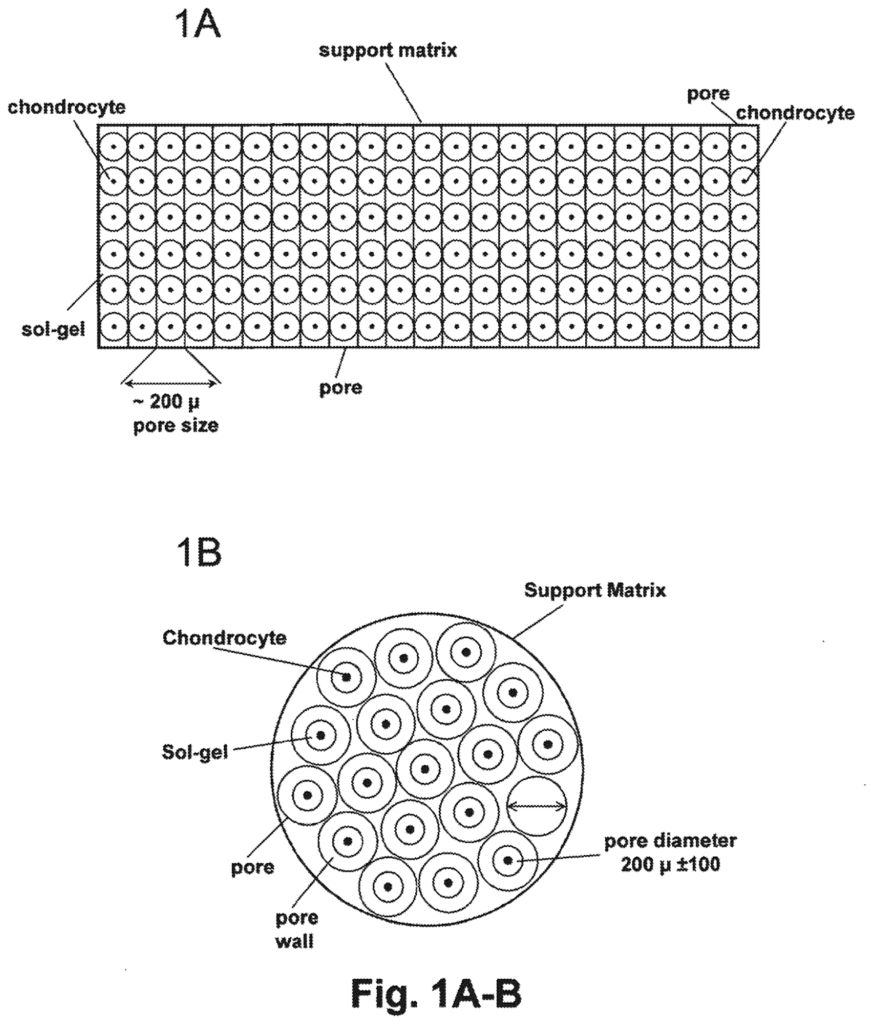
Source – US2025171736A1
Despite the excitement around scaffold-free systems, the patent landscape still shows a field deeply focused on architecture, porosity, mechanical survival, and biological performance.
Another signal that emerged from the analysis was that bioprinting is beginning to fragment into tissue-specific engineering domains.
You see, earlier research often approached bioprinting as a single platform technology. But recent patent activity suggests the field is entering a more mature stage where materials, scaffold designs, and fabrication strategies are being tuned for specific biological targets.
For example, US2025172541A1 focuses on bioinks and fabrication methods designed specifically for bone tissue models. The patent application filed by Texas Tech University aims to create printed constructs that better replicate bone structure and cellular behavior, making them useful for studying bone regeneration and disease.
Meanwhile, CN118048298A by Xiamen University explores myocardial (heart) tissue engineering, describing a method to develop scaffold systems that support cardiac cell organization. Whereas CN118873746A describes bioinks optimized for printing nerve cells, addressing the delicate conditions required for neural tissue formation.
It is clear that bioprinting is now moving towards a tissue-by-tissue engineering discipline, in which each biological target requires its own materials and fabrication strategies.
One of the biggest challenges in bioprinting is not printing cells. It is keeping them alive after printing.
In real tissues, cells survive because blood vessels constantly deliver oxygen and nutrients while removing waste. But when thick tissue constructs are printed in the lab, those internal circulation systems are often missing. Without them, cells in the inner layers quickly die.
This is why vascularization, i.e., the creation of tiny blood vessel networks, has become one of the most important technical challenges in the field.
Thankfully, the patent landscape reflects just how much effort is going into solving this problem.
For example, filings like CN118879611A describe a gradient vascular tissue model, that allows researchers to construct layered vascular structures that closely mimic how real blood vessels are organized in living tissue.
This is just one example. There are several others like US2025101358A1 and US2025230390A1, which focus on building the circulation systems that allow those printed tissues to survive and function.
While the dream of implantable printed organs captures public attention, much of today’s innovation is happening in supporting technologies and experimental systems.
For instance, bioprinted tissue models and organoids are already allowing researchers to simulate organ behavior in controlled laboratory environments, enabling more accurate disease modeling and drug testing.
But these signals are only a part of the picture. The patent landscape around bioprinting is far broader, and many additional directions are emerging across materials science, regenerative medicine, and biofabrication.
By now, we can see that the bioprinting landscape is far richer than it might initially appear, and there is a lot to learn. If your work is in the area of bioprinting, it is a good idea to study the landscape to understand where innovation is happening and where the white spaces might exist.
And to do that, you don’t need to be an expert at writing complex search queries either. With PQAI’s natural language search capabilities, you can explore this technology or any for that matter in far more depth. The AI patent search platform lets you run plain-language technical queries and discover relevant patents quickly.
In fact, PQAI ranked #2 in an independent study evaluating how effectively AI-powered patent search engines retrieve relevant prior art.
With PQAI, you can identify emerging research directions, uncover related inventions, and understand how innovation is evolving across different parts of the bioprinting stack.
If you are exploring innovations in this domain, PQAI can help you map the patent landscape and discover what others are building. You can explore the landscape yourself on PQAI today. Try the tool for free here.
Read Next: Waste Recycling Innovation Trends: 4 Signals Emerging from Patent Filings
At PQAI, we bring clarity to the world of patents. Through storytelling and insight, we simplify inventions so innovators, researchers, and businesses can learn from the past and build the future.
Share

When you hear the term climate monitoring, the picture that usually comes to mind is a network of satellites and machines deployed globally. You imagine

When you hear the term self-healing materials, the first image that usually comes to mind is a scratched surface or a cracked coating that somehow

“I like to think of synthetic biology as liquid alchemy, only instead of transmuting precious metals, you’re synthesizing new biological functionality inside very small channels.”
Dear PQAI Team,
We are pleased to express our support for PQAI and its mission to revolutionize patent searching through open-source, AI-driven solutions.
At [COMPANY NAME], we recognize the importance of accessible and efficient patent tools in fostering innovation and empowering inventors from diverse backgrounds. By supporting PQAI, we aim to contribute to the development of transparent, collaborative, and impactful solutions for the intellectual property community.
We kindly request the addition of [COMPANY NAME] to the official List of Supporters of PQAI.
Sincerely,
[CEO or Equivalent Name]
[Title]
[Company Name]
[Signature]